Jaroslav Šťastnýa, Dominik Vondráčekb, Emmanuel Arriaga-Varelac, d, Jiří Hájekb, *
a Independent scholar, Kosmonautů 359, CZ-460 05 Liberec, Czech Republic
b Department of Entomology, National Museum, Cirkusová 1740, CZ-193 00 Praha 9-Horní Počernice, Czech Republic
c Department of Zoology, Faculty of Science, Charles University, Viničná 7, CZ-128 44 Praha 2, Czech Republic
d Centro de Estudios en Zoología, CUCBA, Universidad de Guadalajara, Km. 15.5 Carretera a Nogales, Las Agujas, Nextipac, 45110 Zapopan, Jalisco, Mexico
*Corresponding author: jiri.hajek@nm.cz (J. Hájek)
Received: 15 February 2020; accepted: 2 June 2020
http://zoobank.org/urn:lsid:zoobank.org:pub:553F3073-E375-47F0-96E5-027265DF1902
Abstract
Platynectes (Agametrus) zoque sp. nov. is described from the state of Oaxaca, Mexico. It is externally similar to the only Central American Platynectes, P. nitens (Sharp) from Panama and Costa Rica, but can be recognized by the shape of the male genitalia. The genetic distance between both species is 6.8% in the cox1 gene fragment. The finding of Platynectes in Mexico extends the known distribution of the genus in the Americas by about 1,300 km to the northwest. Thirteen species of the subgenus Agametrus Sharp are currently known, occurring in the Cordilleras from Chile to Mexico. A list of all species is provided including 10 new combinations resulting from the phylogenetic analysis of Toussaint et al. (2017): Platynectes (Agametrus) boliviensis (Régimbart, 1899), P. (A.) humilis (Sharp, 1882), P. (A.) labratus (Sharp, 1882), P. (A.) nitens (Sharp, 1887), P. (A.) peruvianus (Laporte, 1835), P. (A.) rotundatus (Brinck, 1948) (all from Agametrus); P. (A.) curtulus (Régimbart, 1899), P. (A.) darlingtoni (Guéorguiev, 1971), P. (A.) gaudichaudii (Laporte, 1835), and P. (A.) muelleri (Kirsch, 1865) (all from Leuronectes Sharp). Platynectes (Gueorguievtes) darlingtoni Guéorguiev, 1972, a junior homonym of P. (A.) darlingtoni (Guéorguiev, 1971), is replaced with its junior subjective synonym P. multimaculatus Watts, 1978.
Keywords: Adephaga; Diving beetles; Taxonomy; New combinations; Cytochrome oxidase I; Barcoding
© 2020 Universidad Nacional Autónoma de México, Instituto de Biología. This is an open access article under the CC BY-NC-ND license
El género Platynectes (Coleoptera: Dytiscidae: Agabinae) en Centroamérica, con el descubrimiento de la especie más septentrional en Oaxaca, México, y notas nomenclaturales sobre el subgénero Agametrus
Resumen
Se describe Platynectes (Agametrus) zoque sp. nov. del estado de Oaxaca, México. Esta especie es externamente similar al único Platynectes centroamericano, P. nitens (Sharp) de Panamá y Costa Rica, pero puede diferenciarse por la forma del edeago. La distancia genética del fragmento del gen cox1 entre ambas especies es 6.8%. El descubrimiento de Platynectes en México extiende la distribución conocida del género en América en aproximadamente 1,300 km al noroeste. Trece especies del subgénero Agametrus Sharp se conocen actualmente, distribuidas en las cordilleras desde Chile hasta México. Se proporciona la lista de todas las especies, incluyendo 10 nuevas combinaciones resultantes del análisis filogenético de Toussaint et al. (2017): Platynectes (Agametrus) boliviensis (Régimbart, 1899), P. nitens (Sharp, 1887), P. (A.) peruvianus (Laporte, 1835), P. (A.) rotundatus (Brinck, 1948) (todas de Agametrus); P. (A.) curtulus (Régimbart, 1899), P. (A.) darlingtoni (Guéorguiev, 1971), P. (A.) gaudichaudii (Laporte, 1835), y P. (A.) muelleri (Kirsch, 1865) (todas de Leuronectes Sharp). Platynectes (Gueorguievtes) darlingtoni Guéorguiev, 1972, un homónimo junior de P. (A.) darlingtoni (Guéorguiev, 1971), es remplazado por su sinónimo subjetivo junior P. multimaculatus Watts, 1978.
Palabras clave: Adephaga; Escarabajos buceadores; Taxonomía; Nuevas combinaciones; Citocromo oxidasa I; Código de barras
© 2020 Universidad Nacional Autónoma de México, Instituto de Biología. This is an open access article under the CC BY-NC-ND license
Introduction
The Agabine genus Platynectes Régimbart includes 68 species occurring in the Australian, Neotropical, Oriental and Palearctic regions (Nilsson & Hájek, 2020). Toussaint et al. (2017) published a comprehensive molecular phylogeny of the genus; they considered the former genus Agametrus Sharp to be a subgenus of Platynectes and synonymised it with another Andean genus, Leuronectes Sharp. Therefore, Platynectes species are currently classified in 4 subgenera (for a brief history of their classification, see Hájek et al., 2019), 2 of which are distributed in the Neotropical region: the Cordilleran Agametrus, and Platynectes s. str. from the Guiana Shield, Amazonia and southeastern Brazil. Toussaint et al. (2017), however, did not provide new taxonomic combinations resulting from their analysis (see below).
Twelve Agametrus species were currently known in mountain systems from Chile to Costa Rica. Recent field work by Mexican entomologists in San Miguel Chimalapa municipality, Oaxaca State, Mexico, revealed the presence of another species, which extends the known distribution of Platynectes in the New World by around 1,300 km to the northwest. The aim of the present paper is to formally describe the new species and compare it with the only hitherto known Central American species of Agametrus, P. nitens (Sharp) from Panama and Costa Rica. In addition, a list of all Agametrus species is provided, and 10 formal new combinations are introduced.
Material and methods
In order to elucidate the phylogenetic position of the new species within Platynectes, DNA sequences of the 3’ region of the mitochondrial cytochrome oxidase I gene (cox1) were obtained. The DNA of 2 specimens of Platynectes zoque sp. nov. was isolated. We also extracted the DNA of one specimen of P. nitens (Sharp) from Panama for comparison. In addition, the 5’ (barcode) region of the cox1 gene is provided for all 3 mentioned samples and made available on the Barcode of Life Data Systems (BOLD; Ratnasingham & Hebert, 2007) under the specific process ID (see type and examined material). Other already existing sequences of Agametrus species were downloaded from GenBank and added to the analysed dataset. As an outgroup, several Australian and American taxa of the genus Platynectes were chosen.
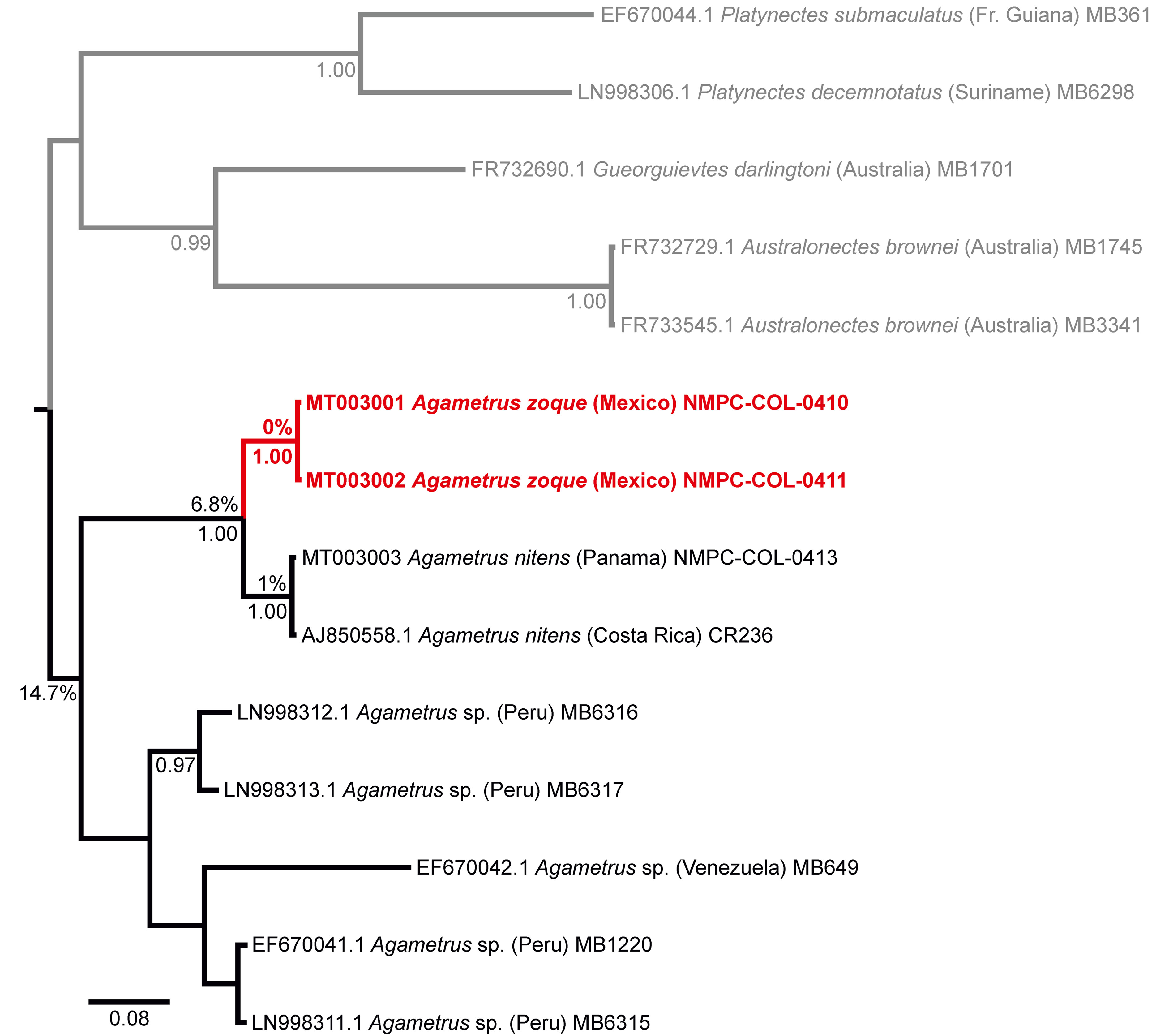
For details about the laboratory protocols, follow Vondráček, Tkoč et al. (2018), including primers and PCR protocol for the barcode region. For the amplification details of the 3’ region of cox1, follow Vondráček, Fuchsová et al. (2018). Obtained sequences of the 3’ region of cox1 were submitted to the GenBank (NCBI) under the accession numbers given in Fig. 1. Methods used for the editing and the subsequent analyses of obtained sequences are described in detail in Šumpich and Jaroš (2019). The only difference was in the number of generations during the Bayesian inference computational method, which was set to 20 X 106 for this study.
Specimens were examined using an Olympus SZX12 stereomicroscope. Habitus photographs were taken using a Canon EOS 550D digital camera with an attached Canon MP-E65mm f/2.8 1-5× macro lens as numerous separate images at different focal planes and afterwards combined using Helicon Focus 6.3.0 software. The male genitalia were studied and illustrated in temporary glycerine mounts using an Olympus BX41 transmitted light microscope with Canon DS126291 attachment; subsequently, they were washed in distilled water and mounted in DMHF on the same card as the beetle.
Measurements were taken with an ocular graticule. The following abbreviations were used in the descriptions: TL – total length of body, a single measurement of length from front of head to apex of elytra; TL-h – total length without head length, length of body from anterior margin of pronotum to apex of elytra; MW – maximum width of body. WC/WS – ratio of width of metacoxa (WC) along extension of line in maximum width of metaventrite (WS) at the point of closest approximation of metacoxa to mesocoxal cavity (Petrov et al., 2010: 43; Fig. 3). The terminology to denote the orientation of the genitalia follows Miller and Nilsson (2003).
For material examined, exact label data are cited and given in quotation marks for type material. Authors’ additional remarks are provided in square brackets. Separate label lines are indicated by a slash (/), separate labels by a double slash (//). The specimens included in this study are deposited in the following collections: BMNH, The natural History Museum [former British Museum (Natural History)], London, United Kingdom (Christine Taylor); CNIN, Colección Nacional de Insectos, Instituto de Biología, Universidad Nacional Autónoma de México, Mexico City, Mexico (Santiago Zaragoza-Caballero); JSCL, Jaroslav Šťastný collection, Liberec, Czech Republic; NMPC, National Museum, Prague, Czech Republic (Jiří Hájek); and ZSMG, Zoologische Staatssammlung, München, Germany (Michael Balke, Lars Hendrich).
Results
The reconstructed phylogenetic tree based on a fragment of the 3’ region of cox1 gene clearly shows the separation of Platynectes zoque sp. nov. from P. nitens with a genetic distance of 6.8% between the 2 clades formed by these 2 species (Fig. 1). The genetic distance between the 2 specimens of P. zoque sp. nov. from the same collecting event is 0%. On the other hand, there is a genetic distance of 1% between P. nitens from Panama and P. nitens from Costa Rica, whose localities are approximately 235 km apart. The closest relatives of these 2 species are highly separated in a different clade with the average genetic distance 14.7%. Based on the literature focused on other dytiscid beetles and encompassing genetic distance data between species (Bergsten, Brilmyer et al., 2012; Hájek et al., 2019), as well as examples from other beetle groups (Szczepański et al., 2018; Vondráček et al., 2018), we consider the distance between P. nitens and P. zoque sp. nov. sufficient to distinguish both as separate species. Although, we cannot eliminate the possibility of the genetic differences by distance effect in our analyses (Bergsten, Bilton et al., 2012), the localities are more than 1,300 km away, we consider the conspecificity of both taxa as quite improbable due to the discontinuity of high altitude Cordillera mountains between Mexico and Costa Rica-Panama. The genetic distances based on the 5’ region of cox1 resulted in a similar way: there is 0% difference between the 2 specimens of P. zoque sp. nov.; the differences between specimens of P. zoque sp. nov. and P. nitens are 7.8% and 7.9% respectively in the barcode region.
Description
Genus Platynectes Régimbart
Platynectes Régimbart, 1879: 454; gender masculine; type species: Agabus decemnotatus Aubé, 1838 by subsequent designation of Guignot (1946).
Subgenus Agametrus Sharp
Agametrus Sharp, 1882: 491; type species: Colymbetes peruvianus Laporte, 1835 by subsequent designation of Guignot (1946).
Leuronectes Sharp, 1882: 491; type species: Colymbetes gaudichaudii Laporte, 1835 by subsequent designation of Guignot (1946); synonymy by Toussaint et al. (2017).
Diagnosis. Modified from Guéorguiev (1971). Habitus in dorsal view elongate oval, lateral outline evenly curved and continuous between pronotum and elytron. Surface shiny, black, at most with yellowish spots on elytra. Sides of pronotum not bordered; posterior angles acute, protruding posteriorly. Prosternal process broad, flat in cross section, with acute apex. Epipleura sharply narrowed at level of abdominal ventrite I, then almost linear to apex. Metacoxal lines absent or present (former Leuronectes).
Taxonomic summary
Distribution. Thirteen species distributed in mountains from Chile to southern Mexico.
Remarks
Toussaint et al. (2017), in a molecular phylogenetic analysis, showed the paraphyly of Platynectes with respect to Agametrus and Leuronectes Sharp. They treated Agametrus as a subgenus of Platynectes and included in it the species previously assigned to Leuronectes; however, they did not establish new combinations for the species. The new combinations appeared in the World catalogue, which is, however, not a formal publication according to ICZN (Nilsson & Hájek, 2020). Therefore, we list here all Agametrus species and establish the formal new combinations.
Platynectes (Agametrus) andinus Guignot, 1958 (Peru)
Platynectes (A.) boliviensis (Régimbart, 1899) comb. nov. (from Agametrus) (Bolivia)
Platynectes (A.) curtulus (Régimbart, 1899) comb. nov. (from Leuronectes) (Argentina)
Platynectes (A.) darlingtoni (Guéorguiev, 1971) comb. nov. (from Leuronectes) (Chile)
Platynectes (A.) gaudichaudii (Laporte, 1835) comb. nov. (from Leuronectes) (Chile)
Platynectes (A.) humilis (Sharp, 1882) comb. nov. (from Agametrus) (Venezuela)
Platynectes (A.) labratus (Sharp, 1882) comb. nov. (from Agametrus) (Venezuela)
Platynectes (A.) monticola Guignot, 1958 (Peru)
Platynectes (A.) muelleri (Kirsch, 1865) comb. nov. (from Leuronectes) (Colombia)
Platynectes (A.) nitens (Sharp, 1887) comb. nov. (from Agametrus) (Panama)
Platynectes (A.) peruvianus (Laporte, 1835) comb. nov. (from Agametrus) (Peru)
Platynectes (A.) rotundatus (Brinck, 1948) comb. nov. (from Agametrus) (Peru)
Platynectes (A.) zoque sp. nov. (Mexico)
As a consequence of new combinations mentioned above, P. (Gueorguievtes) darlingtoni Guéorguiev, 1972 from Australia becomes a junior homonym of P. (A.) darlingtoni (Guéorguiev, 1971). Therefore, the junior name is replaced with its junior subjective synonym P. multimaculatus Watts, 1978, based on synonymy published by Watts (1985).
Platynectes (Agametrus) nitens (Sharp)
Figs. 2a, 3a
Agametrus nitens Sharp, 1887: 756.
Diagnosis. Black, with 1 postmedian (at about 58% of elytral length) lateral vague yellow spot on each elytron (Fig. 2a). Reticulation of dorsal surface weakly impressed, consisting of heterogeneous polygonal meshes; meshes often not closed, usually with larger punctures at interspaces of meshes and minute punctures inside meshes. Ratio WC/WS 3.30-3.50. Metacoxal lines absent. Median lobe of aedeagus in lateral aspect simple, sickle-shaped, uniformly broad throughout its length, distinctly setose apically; apex rounded (Fig. 3a).
Measurements. TL = 6.6-7.7 mm (6.9 ± 0.2 mm), TL-h = 6.2-7.1 mm (6.4 ± 0.2 mm), MW = 3.8-4.3 mm (3.9 ± 0.1 mm).
Taxonomic summary
Type locality. Panama (Ngäbe-Buglé), Peña Blanca (ca. 08°29’ N, 81°38’ W).
Type material. Holotype (male, BMNH): “Agametrus nitens / Type D.S. / Pena Blanca. Panama / 3-4000ft. Champion [Sharp’s handwriting] // Type / H.T. (printed, round label with red margin) // Sharp Coll. / 1905-313. (printed)”.
Additional material examined. Costa Rica, Tapanti NP, ditch by Cabinas at park entrance, 09°45.664’ N, 83°47.091’ W, 1,200 m, 8-Jan-2005 , A. Short, J. Hannam, A. Swanson leg. (2 specimens, ZSMG); Panama, Chiriqui Prov., Cordillera Talamanca Mts., Volcán Ciudad, Las Tierras Altas, 08°50.591’ N, 82°41.483’ W, 598 m, Apr-May-2014, M. Häckel leg. (1 male, NMPC); Panama, Chiriqui, Boquete, Lost Waterfalls Trail, 08°50.5’ N, 82°28.6’ W, 1,750-1,900 m, montane forest, pools, 8-Sep-2017, Fikáček, Hájek, Seidel & Sekerka leg. (24 specimens, JSCL and NMPC; 1 specimen sequenced, with DNA voucher number: NMPC-COL-0413, and BOLD Process ID: DNPMC662-20; Panama, Ngäbe-Buglé, Serranía de Tabasara, 22-Sep-2014, M. Veselý leg. (1 male, 2 females, JSCL).
Collecting circumstances. In Chiriqui, the specimens were collected in remnant pools of forest streamlet (Fig. 4).
Distribution. Costa Rica (Blanco-Aller & Régil, 2016) and western Panama (Chiriqui, Ngäbe-Buglé).
Platynectes (Agametrus) zoque sp. nov.
Figs. 2b, 3b, c
http://zoobank.org/urn:lsid:zoobank.org:act:2A9E5294-
6664-42AF-835A-9134695D776B
Diagnosis. The new species is externally similar to P. nitens, but it can be recognized by its slightly greater body length. The undoubtful identification is only possible based on the study of the male genitalia: the median lobe of P. zoque sp. nov. in lateral view is slender in its middle part, and distinctly broadened apically.

Description (male holotype). Habitus in dorsal view elongate oval, rather narrow, broadest before elytral midlength (Fig. 2b); lateral outline evenly curved and continuous between pronotum and elytron. Entire surface shiny. Coloration: dorsal surface black with reddish-brown marking: head black, 2 posteromedial oval spots on vertex and anterior margin of clypeus reddish brown; labrum brownish. Pronotum black, anterior corners and a vague tranverse area on disk reddish brown; margins indistinctly reddish-brown translucent. Elytra black, with 1 postmedian (at about 58% of elytral length) lateral vague yellow spot on each elytron. Ventral surface black, epipleura dark reddish-brown; prosternal process, anterior margin of “metasternal wings” and metacoxae pale reddish brown; abdominal ventrites II-III laterally with a vague reddish spot, ventrite VI pale apically. Appendages yellowish brown. Head: moderately broad, ca 0.65× width of pronotum, transversely elliptical. Anterior margin of clypeus truncate. Antennae with antennomeres moderately broad, club-shaped; antennomere II short, 1.8× longer than wide; antennomeres III-V of equal length, 2.2× longer than wide; antennomeres from VI to XI becoming progressively shorter. Head reticulation simple, consisting of weakly impressed, more or less polygonal meshes of irregular size and shape; meshes mostly closed, usually with several micropunctures inside; meshes dense and smaller along eyes, disappearing on clypeus. Punctation double: coarse setigerous punctures present in 2 small and shallow frontoclypeal grooves, and in depressions along inner margin of eyes; fine punctures distributed mostly in intersections of meshes. Pronotum: transverse, broadest at posterior angles. Anterior angles acute, posterior angles obtusely rectangular. Sides slightly and evenly curved, without distinct lateral beading. Anterior margin almost straight (dorso-frontal view), posterior margin slightly sinuate. Punctation double, similar to that of head; row of coarse imperceptibly setigerous punctures present along anterior and posterior margin; in posterior 2 thirds parallel to lateral margin with irregular row of punctures forming shallow impression posteriad; punctures along anterior margin slightly sparser medially; posterior row of punctures broadly interrupted medially, but not interrupted sublaterally. Fine punctures distributed irregularly on pronotal surface, presenting mostly in intersections of reticulation. Reticulation similar to that of head, consisting of heterogeneous polygonal meshes, usually with several micropuctures inside; meshes larger, shallowly impressed and open on disc, becoming more often closed, smaller and deeply impressed near sides. Centre of pronotal disc with indistinct longitudinal furrow. Lateral margins not bordered. Scutellar shield: broadly triangular. Elytra: sides evenly rounded, apically slightly angulate; lateral margin distinctly bordered (lateral epipleural carina distinctly visible throughout length). Punctation double; coarse punctures present in 2 relatively distinct longitudinal discal lines and irregular lateral line, few punctures present also along suture; fine punctures distributed irregularly over elytral surface, occurring mostly in intersections of reticulation. Reticulation finely impressed, similar to that of head and pronotum, consisting of heterogeneous polygonal meshes; meshes often not closed, usually with several micropunctures inside. Legs: meso- and metafemora with bunch of spiniform setae along posterolateral margin. Pro- and mesotibia widened, club shaped, densely punctured with spinigerous punctures over ventral surface. Metatibia with 2 lines of coarse spinigerous punctures over ventral surface.

Pro- and mesotarsomeres I-III moderately dilated, ventrally with transverse rows of oval adhesive setae; protarsal claws equal in length, 0.75× as long as protarsomere V. Metatarsal claws subequal; anterior (lateral) claw slightly shorter and arcuate, posterior (medial) claw longer and straight. Surface of legs with distinct reticulation consisting of elongate oblique or transverse meshes. Elongate natatorial setae present along dorsal margin of all tibiae, and pro- and mesotarsomeres, and along both dorsal and ventral margins (especially dorsal margin) of metatarsomeres. Ventral surface: genae reticulate with transverse meshes. Prosternum sinuate anteriorly, obtusely keeled medially; lateral portions of prosternum transversely reticulate; prosternal column shiny, with sparse double punctation; slopes of prosternal column densely and coarsely punctate; prosternal process moderately lanceolate, almost flat in cross section, pointed at apex, sides broadly beaded except on apex, sparsely and finely punctate. Medial part of metaventrite without microsculpture, shiny, with sparse fine punctation; lateral parts of metaventrite (“metasternal wings”) slender, transversally reticulated, relatively broad. Ratio WC/WS = 3.25. Metaventrite with anteromedian impression almost attaining level of posterior margin of mesocoxae. Metacoxal lines absent. Metacoxal plates reticulated laterally with longitudinally elongated meshes; punctation very fine, sparse. Abdomen: abdominal ventrites I-V with reticulation consisting of longitudinal (I), oblique (II-III) and transverse (IV-V) meshes; punctation double; group of coarse setigerous punctures present in centre of ventrites III-V, additional setigerous punctures arranged sparsely in medial part of ventrites; fine punctures distributed sparsely and irregularly on surface of ventrites. Apical abdominal ventrite (VI) with posterior margin rounded, apically slightly truncate; distinctly beaded, lateral beading continuous, reaching lateral angles; transverse reticulation distinct basolaterally; with long and deep longitudinal grooves; punctation double, coarsely punctured along posterior margin, sparsely and finely punctured on disc. Male genitalia: median lobe in lateral aspect simple, sickle-shaped, only slightly curved, slender in median part and broadened apically (Fig. 3b); apex expanded, broadly rounded and distinctly setose. Parameres slender, triangular with a distinct subbasal tooth on ventral side, apical third narrow and elongate; densely setose dorsally (Fig. 3c).
Female. Differs from male in the following characters: meshes of dorsal surface reticulation slightly more deeply engraved, mostly closed; abdominal ventrite VI with grooves reduced to a series of setigerous punctures; pro- and mesotarsomeres I-III not dilated and without adhesive setae.
Measurements. TL: 7.4-7.6 mm (holotype: 7.6 mm). TL-h: 6.8-6.9 mm (holotype: 6.9 mm). MW: 4.2-4.4 mm (holotype: 4.4 mm).
Taxonomic summary
Type locality. Mexico, Oaxaca State, San Miguel Chimalapa Municipality, San Antonio, Paraje El Gringo, ca. 16°40’53.39” N, 94°14’0.2” W, 1,796 m asl.
Type material. Holotype male (CNIN): “Mexico, Oaxaca, San Miguel Chimalapa / San Antonio “Paraje El Gringo” / Zone of “Paraje El Retén” (forest pond) / 16°40’53.39” N, 94°14’0.2” W / 1,796 m, 23-26-Jul-2017 / Arriaga, Alvarado, Ponce et al. lgt.” [printed] // holotype ♂ / Platynectes / zoque sp. nov. / J. Šťastný & J. Hájek det. 2019 [red label, printed]”. Paratypes (1 male, 3 females), same label data as holotype; 2 females with additional printed DNA voucher number: p “NMPC-COL-410” [411, respectively], and with BOLD Process ID: “BOLD DNMPC660-20” [DNMPC661-20 respectively] (CNIN, NMPC); each paratype is provided with the appropriate red printed label.
Etymology. The new species is named in honour of Zoque tribe —an indigenous people in Mexico, living in the area of San Antonio. The name is a noun in the nominative case.
Collecting circumstances. The type specimens were collected in a forest pool formed by an intermittent stream in a cloud forest.
Distribution. So far known only from the type locality in southwestern Oaxaca State, southern Mexico.

Acknowledgements
We are obliged to Hans Fery (Berlin, Germany) for information about the homonymy of P. darlingtoni, to Robert B. Angus (Natural History Museum, London, UK), who improved the language of the manuscript, and to two anonymous reviewers for valuable comments on the manuscript. The present work was supported by the Ministry of Culture of the Czech Republic (DKRVO 2019-2023/5.I.b, National Museum, 00023272).
References
Bergsten, J., Bilton, D. T., Fujisawa, T., Elliott, M., Monaghan, M. T., Balke, M. et al. (2012). The effect of geographical scale of sampling on DNA barcoding. Systematic Biology, 61, 851–869. https://doi.org/10.1093/sysbio/sys037
Bergsten, J., Brilmyer, G., Crampton-Platt, A., & Nilsson, A. N. (2012). Sympatry and colour variation disguised well-differentiated sister species: Suphrodytes revised with integrative taxonomy including 5 kbp of housekeeping genes (Coleoptera: Dytiscidae). DNA Barcodes, 1, 1–18. https://doi.org/10.2478/dna-2012-0001
Blanco-Aller, R., & Régil, J. A. (2016) Los géneros Agametrus Sharp, 1882 y Rhantus Dejean, 1833 (Coleoptera: Adephaga: Dytiscidae) en Costa Rica: composición faunística y nuevas citas. Boletín de la Sociedad Entomológica Aragonesa, 58, 123–129.
Guéorguiev, V. B. (1971). Notes sur les Agabini (Coleoptera, Dytiscidae). I. Les genres Agametrus Sharp, Leuronectes Sharp et Andonectes gen. n. Izvestija na Zoologitjeskija Institut s Musei Sofia, 33, 165–176.
Guignot, F. (1946). Génotypes des Dytiscoidea et des Gyrinoidea. Revue Française d’Entomologie, 13, 112–118.
Hájek, J., Alarie, Y., Šťastný, J., & Vondráček, D. (2019). The first hygropetric Platynectes and its larva from eastern China (Coleoptera: Dytiscidae). Acta Entomologica Musei Nationalis Pragae, 59, 217–228. https://doi.org/10.2478/aemnp-2019-0019
Miller, K. B., & Nilsson, A. N. (2003). Homology and terminology: communicating information about rotated structures in water beetles. Latissimus, 17, 1–4.
Nilsson, A. N., & Hájek, J. (2020). A world catalogue of the family Dytiscidae, or the diving beetles (Coleoptera, Adephaga) Version 1.I.2020. Available: http://www.waterbeetles.eu
Petrov, P. N., Shapovalov, M. I., & Fery, H. (2010). A new species of Ilybius Erichson, 1832 (Coleoptera: Dytiscidae) from the North Caucasus. Russian Entomological Journal, 19, 41–45.
Ratnasingham, S., & Hebert, P. D. N. (2007). BOLD: The Barcode of Life Data System (http://www.barcodinglife.org). Molecular Ecology Notes, 7, 355–364. https://doi.org/10.1111/j.1471-8286.2007.01678.x
Régimbart, M. (1879). Étude sur la classification des Dytiscidae. Annales de la Société Entomologique de France, Cinquième Série, 8, 447–466.
Sharp, D. (1882) On aquatic carnivorous Coleoptera or Dytiscidae. Scientific Transactions of the Royal Dublin Society, Series II, 2, 179–1003.
Šumpich, J., & Jaroš, J. (2019). Chrysoclista karsholti, sp. n., from Turkey, and a new record of C. germanica from central Europe (Lepidoptera: Elachistidae: Parametriotinae). Zootaxa, 4568, 587–592. https://doi.org/10.11646/zootaxa.4568.3.12
Szczepański, W. T., Vondráček, D., Seidel, M., Wardhaugh, C., & Fikáček, M. (2018). High diversity of Cetiocyon beetles (Coleoptera: Hydrophilidae) along an elevational gradient on Mt. Wilhelm, New Guinea, with new records from the Bird’s Head Peninsula. Arthropod Systematics & Phylogeny, 76, 323–347.
Toussaint, E. F. A., Hendrich, L., Hájek, J., Michat, M. C., Panjaitan, R., Short, A. E. Z. et al. (2017). Evolution of Pacific Rim diving beetles sheds light on Amphi-Pacific biogeography. Ecography, 40, 500–510. https://doi.org/10.1111/ecog.02195
Vondráček, D., Fuchsová, A., Ahrens, D., Král, D., & Šípek, P. (2018). Phylogeography and DNA-based species delimitation provide insight into the taxonomy of the polymorphic rose chafer Protaetia (Potosia) cuprea species complex (Coleoptera: Scarabaeidae: Cetoniinae) in the Western Palearctic. Plos One, 13, e0192349. https://doi.org/10.1371/journal.pone.0192349
Vondráček, D., Tkoč, M., & Fikáček, M. (2018). Is repeated cypermethrin fumigation dangerous for the mitochondrial DNA in dry insect samples? Acta Entomologica Musei Nationalis Pragae, 58, 609–614. https://doi.org/10.2478/aemnp-2018-0051
Watts, C. H. S. (1985). A faunal assessment of Australian Hydradephaga. Proceedings of the Academy of Natural Sciences Philadelphia, 137, 22–28.




