Ronald F. Jesus, Evelyn L. Cardoso, Yuri Willkens, Adriano P. Furtado,
Jeannie N. dos Santos, Francisco T. V. Melo *
Laboratório de Biologia Celular e Helmintologia “Profa Dra Reinalda Marisa Lanfredi”, Instituto de Ciências Biológicas, Universidade Federal do Pará, Av. Augusto Corrêa 01 – Guamá, 66075-110 Belém, Pará, Brazil
*Corresponding author: ftiago@ufpa.br (F.T.V. Melo)
Received: 03 April 2020; accepted: 25 May 2020
Abstract
During a helminthological survey on freshwater turtles from the Brazilian Amazon, we collected fresh and well-preserved nematodes from the stomach of Podocnemis unifilis Troschel, 1848, that resembled Orientatractis leiperi. The morphology and morphometry of specimens described herein fits those from the original description. We also observed important morphological characters not reported previously or not properly described, and those are reported here for the first time: the structure of the excretory pore (surrounded by radial cuticle striations), structure and position of deirids, phasmids, oral papillae and the presence of a dorsolateral pair of papilla in caudal region. The present work provides a detailed redescription for O. leiperi, 50 years after its original description, adding a new host and locality for this species.
Keywords: Turtle parasites; Brazilian Amazon; Atractidae; Orientatractis leiperi
© 2020 Universidad Nacional Autónoma de México, Instituto de Biología. This is an open access article under the CC BY-NC-ND license
“Largo tiempo sin verte”: redescripción de Orientatractis leiperi
(Nematoda: Atractidae) parásito de Podocnemis unifilis (Testudines: Podocnemididae) del estado de Pará, Brasil
Resumen
Durante el análisis helmintológico de tortugas de agua dulce en la Amazonía brasileña, fueron extraídos del estómago de Podocnemis unifilis Troschel, 1848, nemátodos que parecían pertenecer a la especie Orientatractis leiperi; estos ejemplares fueron utilizados para realizar la redescripción de esta especie mediante microscopía óptica y microscopía electrónica de barrido. La morfología y morfometría de los ejemplares analizados en este estudio se ajustaron a la descripción original. Importantes caracteres morfológicos que no fueron considerados previamente o que no fueron descritos adecuadamente, se detallan por primera vez en este trabajo: la estructura del poro excretor (rodeado de estrías radiales cuticulares), estructura y posición de las papilas cervicales, fasmidios y papilas orales, distribución y disposición de las papilas caudales en los machos y la presencia de un par de papilas dorsolaterales en la región caudal. Este trabajo presenta una redescripción detallada de O. leiperi, 50 años después de su descripción original, adicionando un nuevo hospedador y nueva localidad para la misma.
Palabras clave: Parásitos de tortugas; Amazonía brasileña; Atractidae; Orientatractis leiperi
© 2020 Universidad Nacional Autónoma de México, Instituto de Biología. This is an open access article under the CC BY-NC-ND license
Introduction
The family Atractidae Railliet, 1917, includes 26 genera of intestinal parasitic nematodes of fish, amphibians and reptiles. Among those genera, Atractis Dujardin, 1845; Buckleyatractis Khalil & Gibbons, 1988; Klossinemella Costa, 1961; Orientatractis Petter, 1966; Paraorientatractis Gibbons, Khalil & Marinkelle, 1997; Paratractis Sarmiento, 1959; Pneumoatractis Bursey, Reavill & Greiner, 2009; Podocnematractis Gibbons, Khalil & Marinkelle, 1995; and Rhinoclemmysnema Gibbons & Platt, 2006, are commonly found as parasites of freshwater turtles (Berenguer, 1947; Bursey et al., 2009; Costa et al., 1968; Gibbons & Platt, 2006; Gibbons et al., 1995, 1997; Khalil & Gibbons, 1988; Petter, 1966; Sarmiento, 1959).
The genus Orientatractis was erected to accommodate Orientatractis levanhoai Petter, 1966 of Indotestudo elongata (Blyth, 1854) from Vietnam, that showed unique shape, number and disposition of sclerotized structures surrounding the oral opening (Petter, 1966). Until now, 10 valid species are known for this genus: Orientatractis asymmetrica Gibbons & Platt, 2006; Orientatractis brycini González-Solís & Mariaux, 2017; Orientatractis campechensis González-Solís & Moravec, 2004; Orientatractis chiapasensis González-Solís & Moravec, 2004; Orientatractis hamabatrachos Bursey, Goldberg & Kraus, 2014; Orientatractis leiperi Buckley, 1969; Orientatractis levanhoai Petter, 1966 (type-species of the genus); Orientatractis longicaudata Liu, Li & Zhang, 2018; Orientatractis mekongensis Moravec, Kamchoo & Pachanawan, 2015; and Orientatractis moraveci Cavalcante, Silva, Santos, Chagas-Moutinho & Santos, 2017 (Buckley, 1969; Bursey et al., 2014; Cavalcante et al., 2017; Gibbons & Platt, 2006; González-Solís & Mariaux, 2017; González-Solís & Moravec, 2004; Liu et al., 2018; Moravec et al., 2015; Petter, 1966).
During a helminthological survey of parasites of freshwater turtles from the Amazon region, we collected and examined parasites of Podocnemis unifilis Troschel, 1848. The turtles harbored countless nematodes in the stomach, which belong to O. leiperi that has not been reported since its original description (51 years ago). Buckley (1969) mentioned that specimens found in the intestines of Podocnemis vogli Müeller, 1935 from Colombia were partially decomposed, especially the internal organs such as the ovary and the uterus which in consequence could not be described or illustrated adequately. Thus, the redescription of O. leiperi is herein presented, adding new morphometric data, new host, and locality for the species.
Materials and methods
Two specimens of P. unifilis were collected from Tocantins River, Tucuruí municipality, Pará, Brazil (8º5’8.797” S, 48º6’48.78” W) (license permission SISBIO: 53527-4). Hosts were anesthetized using Ketamine 2% injection and posteriorly euthanized. Internal organs were removed and examined under a LEICA EZ4 stereo-microscope. Nematodes collected were washed in 0.9% saline solution and fixed in A.F.A solution (2% glacial acetic acid, 3% formaldehyde, and 95% ethanol 70 ºGL). For morphological and morphometric analyses, specimens were cleared in Aman’s Lactophenol, mounted on temporary slides and examined under an Olympus BX41 light microscope with a drawing tube attached.
For scanning electron microscopy, some specimens were post-fixed in 1% OsO4, dehydrated in ethanol series, critical-point dried, coated with gold-paladium, mounted in stubs and analyzed under a Vega3 Microscope (TESCAN, Brno, Czech Republic) at the Laboratory of Embryology and Histology of the Federal Rural University of Amazonia.
Measurements are shown in micrometers, unless otherwise stated. We tried to access the type material deposited in the London School of Hygiene and Tropical Medicine, however they were missing or absent. We also checked the collection of the Natural History Museum of London, in case of transference of the original collection or donation of specimens, but we could not find the type series. Given the significance of these findings, the neotypes have been deposited in the Helminthological Collection of Oswaldo Cruz Institute, Brazil, one of the most important parasite collections in the country and has an important tradition in parasite studies world-wide.
Redescription
Family Atractidae (Railliet, 1917 subfam.) Travassos, 1919
Genus Orientatractis Petter, 1966
Orientatractis leiperi Buckley, 1969 (Figs. 1, 2)
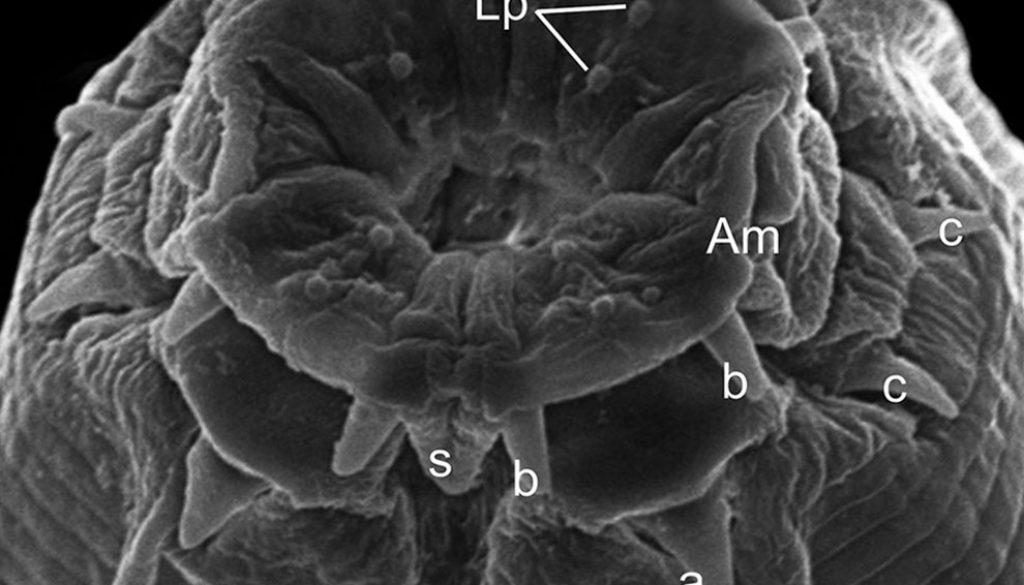
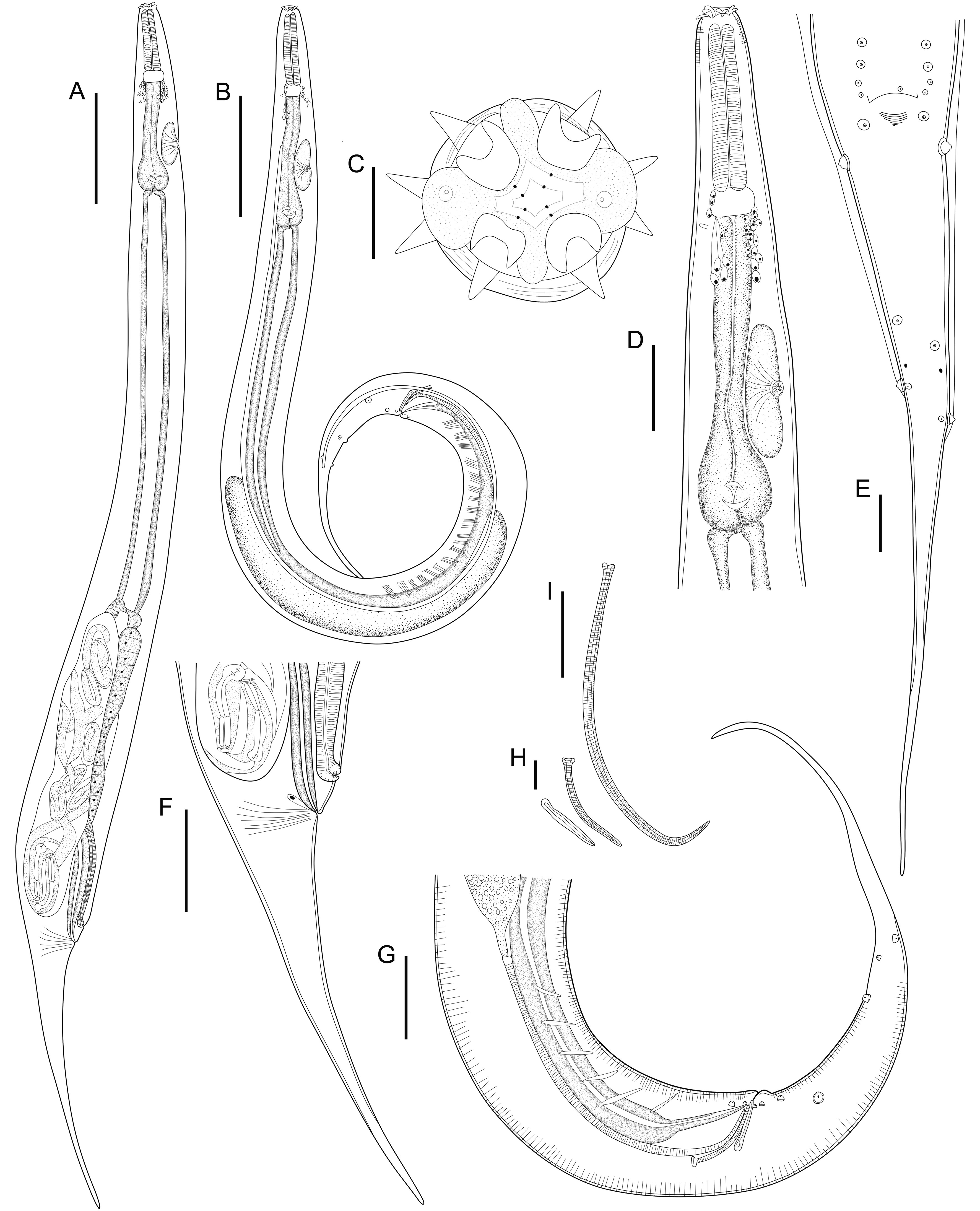
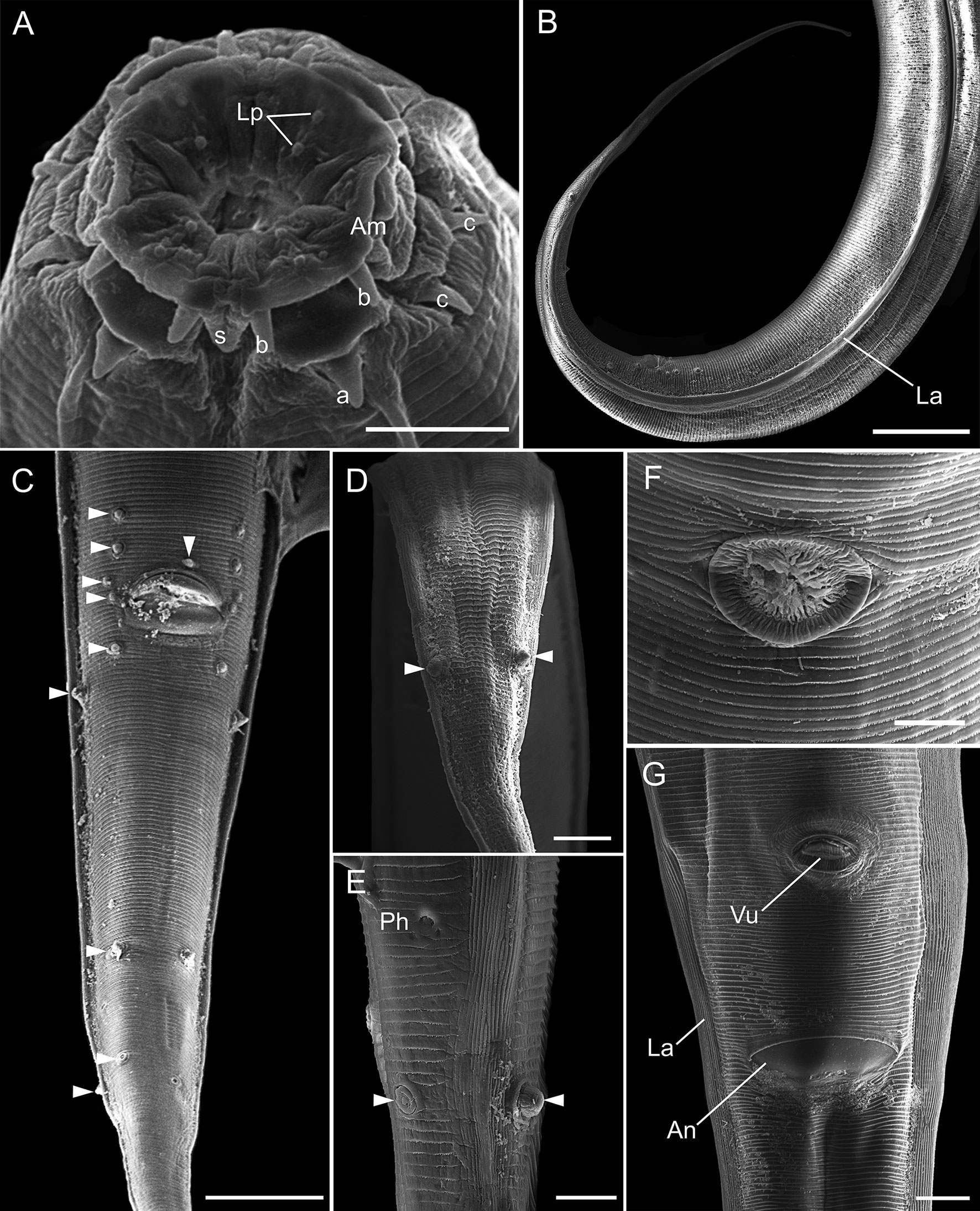
General. Small and slender nematodes, with cuticle finely transversely striated. Anterior end rounded (Fig. 1A, B). Lateral alae well developed starting from first third of esophagus, ending at dorsolateral pair of cauda papillae in posterior extremity of body (Figs. 1B, E, 2B, C). Oral aperture irregular, hexagonal to circular, surrounded by 8 submedian papillae in 2 circles, each with 4 papillae. Six lips: 4 submedian (2 subdorsal and 2 subventral) each lip bearing a well-sclerotized, recurved, pointed spines joined at the base and a single large median spine. Two lateral lips bearing amphidial pores and a well-sclerotized U-shaped structure, small spines posterior to each amphidial pore (Figs. 1C, 2A). Esophagus divided into muscular anterior portion and glandular posterior portion ending in a well-developed bulb with sclerotized valves. Nerve ring situated at anterior portion of esophagus, just after muscular portion (Fig. 1A, B, D). Deirids small, located at level of nerve ring. Excretory pore slightly anterior to esophageal bulb, aperture surrounded by radial cuticle striations, pore opening in a large chamber with transversal grooves (Figs. 1A, B, D, 2F). Posterior end with very slender, long, pointed tail (Figs. 1A, B, E, F, G, 2B).
Male (Based on neotype; measurements of 11 paraneotypes in parentheses). Total length 3.21 (3.12-3.37) mm, width at esophago-intestinal junction 152 (107-168). Lateral alae beginning 285 (237-285) from anterior end of body, ending 485 (360-485) from posterior end of body. Esophagus 632 (571-776) long, muscular portion 253 (181-253), glandular portion including esophageal bulb 379 (352-595). Esophageal bulb 107 (96-125) × 93 (64-96). Nerve ring, excretory pore and deirids 280 (203-280), 499 (435-499) and 256 (213-256), respectively, from anterior end of body. Nine pairs of caudal papillae plus 1 unpaired anterior to cloacal lip, of which 3 subventral precloacal pairs, 1 subventral adcloacal pair and 5 postcloacal pairs (3 subventral, 1 ventrolateral and 1 dorsolateral) (Figs. 1E, G, 2C-E). Phasmids located between pairs 4 and 5 of postcloacals papillae (Figs. 1E, 2E). Spicules unequal, lanceolate, proximal ends slightly expanded, distal ends sharply pointed, with transverse striations along their lengths, left spicule 472 (416-491), right spicule 103 (90-119) (Fig. 1I). Gubernaculum with proximal end expanded, deep medial depression, distal end pointed, 64 (58-69) long (Fig. 1H). Tail long, pointed, ventrally curved, 739 (648-781) long (Figs. 1B, E, G, 2B).
Females (Based on neotype female; measurements of 11 paraneotypes in parentheses). Total length 3.64 (3.31-3.68) mm, width at esophago-intestinal junction 160 (128-187). Lateral alae beginning 253 (219-253) from anterior end of body, ending 774 (721-847) from posterior end of body. Esophagus 590 (525-619) long, muscular portion 187 (171-216), glandular portion including esophageal bulb 403 (352-432). Esophageal bulb 99 (80-133) × 120 (101-131). Nerve ring, excretory pore and deirids 208 (179-232), 453 (387-488) and 221 (213-261), respectively, from anterior end of body. Viviparous, vulva aperture surrounded by radial cuticle striations with posterior lip slightly elevated, situated 0.96 (0.96-1.08) mm from posterior end of body (Fig. 2G), anterior to anal opening (Fig. 1A, F). Distance anus-vulva 99 (93-109). Vagina muscular, first portion anteriorly directed, flexed posteriorly. Ovary directed anteriorly to vagina. Monodelphic, prodelphic, uterus beginning in posterior flexion of vagina, directing anteriorly, containing fully developed larvae. Larvae total length 363 and 334 (n = 2, measured in uteri of neotype; Fig. 1A). Eggs 171-139 × 64-51 (n = 7, measured in uteri of neotype). Tail subulate, 900 (874-989) long (Fig. 1A, F).
Taxonomic Summary
Neotype host: Podocnemis unifilis Troschel, 1848.
Neotype locality: Rio Tocantins Tucuruí, Pará, Brazil (8º5’8.797” S, 48º6’48.78” W).
Type-specimens: neotype male [here designed] (CHIOC 38765a), neotype female (CHIOC 38765b) and 22 paraneotypes (CHIOC 38765c-38765d) in the Helminthological Collection of Oswaldo Cruz Institute – Rio de Janeiro, Brazil.
Site of infection: stomach.
Prevalence: 2 infected/2 examined hosts; intensity of infection: countless nematodes per turtle.
Remarks
Here we redescribe Orientatractis leiperi based on specimens found in the stomach of P. unifilis from the Brazilian Amazon Region. The original description of O. leiperi was based on specimens collected from Podocnemis vogli sent to Buckley by Dr. C. J. Marinkelle from Colombia. The neotype described here was found in the same biogeographical region (Neotropical Region, Tucuruí, Pará, Brazil) and ecological biome (Amazon Region), parasitizing a species of host from the same genus (Podocnemis unifilis).
The specimens were allocated to this genus by the presence of 4 single horns surrounding the mouth. According to González-Solís and Moravec (2004), Orientatractis and Klossinemella Costa, 1961, are morphologically similar genera, differing by the number and disposition of sclerotized structures surrounding the oral opening: Klossinemella spp. are characterized by the presence of 8 Y-shape well-sclerotized with 2 pointed tips and 4 single horns, while Orientatractis have 4 bicornuate pieces and 4 single horns.
The morphological characteristics used to differentiate the species of this genus are number and distribution of caudal papillae shape and size of spicules, gubernaculum and morphology and length of tail. Our specimens have 9 pairs of caudal papillae and among the 10 valid species of the genus, O. asymmetrica, O. campechensis, O. chiapasensis and O. leiperi share this morphological trait. However, the specimens found in the present work can be differentiated from O. asymmetrica because the last species have a pair of double papillae at the precloacal region and enlarged on the right side (not observed in our specimens). Additionally, the specimens of the present work have larger spicules (left 416-491 and right 90-119 spicules vs. left 220-239 and right 86-104 spicules in O. asymmetrica) and the presence of a dorsal caudal papillae, which is absent in O. asymmetrica.
The specimens described herein, resembles O. campechensis by the number of cloacal papillae and the size of spicules and gubernaculum. However, our specimens have a gubernaculum with proximal end expanded, deep medial depression and pointed distal end, while O. campechensis have a gubernaculum with a proximal end showing a deep depression forming 2 branches and a dorsal pointed barb, and the females of O. campechensis have a vulvar appendix or elevated vulvar lips.
Orientatractis leiperi found in the stomach of Podocnemis unifilis are similar to O. chiapasensis by number of cloacal papillae, however O. leiperi differ by the number of post cloacal papilla (5 vs. 4 in O. chiapasensis), larger body length (males 3.12-3.37 mm, females 3.31-3.68 mm vs. males 2.709-2.967 mm, females 2.025-3.800 mm in O. chiapasensis), larger spicules (left spicule 416-491, right spicule 90-119 vs. left spicule 204-238, right spicule, 60-68 in O. chiapasensis).
Thus, our specimens can be allocated to O. leiperi by the combination of the following characters: number and distribution of cloacal papillae, size and shape of spicules and gubernaculum and tail length. The specimens of O. leiperi from P. unifilis did not show a substantial intraspecific variation of the metrical data, especially when compared with the measurements of the original description. In our specimens, males are smaller than O. leiperi described by Buckley (1969) (3.12-3.37 vs. 3.80), we also observed differences in total length of esophagus in females (525-619 vs. 660 in original description) and right spicules and gubernaculum of males are smaller in our specimens (right spicule: 90-119 vs. 160, gubernaculum 58-69 vs. 76). However, morphometric variability always occurs intraspecifically, which according to González-Solís and Moravec, (2004) might be associated with local ecological conditions and physiological traits of hosts.
According to Buckley (1969), the description of O. leiperi was based in specimens partially deteriorated, thus the description and illustration of the uterus was incomplete; moreover, the author found embryonated eggs in only one female. Since the type material seems to be lost, our specimens are herein considered as neotypes, which fit the original description by Buckley (1969). Fresh specimens allowed us to verify that females are monodelphic, prodelphic. Additionally, our specimens harbored eggs and fully developed larva in uterus.
The life cycle of Orientatractis spp. is still unknown, however according to studies done in other Atractidae members, these nematodes are viviparous, and larvae develop directly in uterus allowing the auto infection (Anderson, 2000). We found countless specimens of O. leiperi in the stomach of P. unifilis and the viviparity can considerably contribute with the development of adult nematodes directly in the infected organ, increasing the abundance of infection. Additionally, according to Leão et al. (2019), the zones of intensive use of habitat are usually overlapped among specimens of P. unifilis, where the turtles share areas for feeding, copulating, basking, and parental care, which can contribute to the transmission of this species of nematodes.
Our SEM analyses were important to observe the complexity of oral aperture and cephalic papillae, lateral alae, structure of excretory pore, vulva, anus and structures not described by Buckley (1969), such as the presence of dorsolateral caudal papillae in males, phasmids, and cephalic papillae.
This study is the first report of O. leiperi in the Brazilian Amazon region, in a different host species, and encountered after 50 years since its original description.
Acknowledgements
To Missiene Maria da Silva for her support in host necropsies and collection of nematodes; to Márcio André da Silva Dias and Raiel Correia de Miranda for helping with line drawings composition; to Elane Guerreiro Giese, from the Laboratory of Animal Embriology and Histology from the Universidade Federal da Amazônia, Belém, for her technical support with the SEM analyses. This work was supported by CAPES/PPGBAIP/UFPA the National Council for Scientific and Technological Development (CNPq) (grant number 431809/2018-6 Universal); CNPq Research grant productivity to MELO, F. T. V. (Process Nº 304955/2018-3) and SANTOS, J. N. (Process Nº 305552/2019-8). This study is part of the master’s dissertation of Ronald Ferreira de Jesus in the Graduate Program in Biology of Infectious and Parasitic Agents [Programa de Pós-Graduacão em Biologia de Agentes Infecciosos e Parasitários] ICB-UFPA.
References
Anderson, R. C. (2000). Nematode parasites of vertebrates. Their development and transmission. Wallingford, Oxon: CABI Publishing.
Berenger, J. G. (1947). Revisión de la familia Atractidae Travassos, 1920 con descripción de dos nuevas especies. Revista Ibérica de Parasitología, 7, 3–90.
Buckley, J. J. C. (1969). On a remarkable oxyurid nematode, Orientatractis leiperi n. sp., (Atractidae) from a South American tortoise, Podocnemis vogli. Journal of Helminthology, 43, 281–286. https://doi.org/10.1017/S0022149X0000482X
Bursey, C. R., Goldberg, S. R., & Kraus, F. (2014). New species of Orientatractis (Nematoda: Atractidae), new species of Rondonia (Nematoda: Atractidae) and other helminths in Austrochaperina basipalmata (Anura: Microhylidae) from Papua New Guinea. Acta Parasitologica, 59, 115–121. https://doi.org/10.2478/s11686-014-0217-3
Bursey, C. R., Reavill, D., & Greiner, E. (2009). Pneumoatractis podocnemis n. gen., n. sp. (Nematoda: Atractidae) from the Yellow-Spotted Amazon River Turtle, Podocnemis unifilis (Testudines: Pelomedusidae). Comparative Parasitology, 76, 149–153. https://doi.org/10.1654/4379.1
Cavalcante, P. H. O., Silva, M. T., Santos, E. G. N., Chagas-Moutinho, V. A., & Santos, C. P. (2017). Orientatractis moraveci n. sp. and Rondonia rondoni Travassos, 1920 (Nematoda: Atractidae), parasites of Pimelodus blochii (Osteichthyes, Pimelodidae) from the Acre and Xapuri Rivers, Western Amazon, Brazil. Parasitology, 144, 1–11. https://doi.org/10.1017/S0031182016001736
Costa, S. C. G., Motta, C. S., & Gomes, D. C. (1968). Revisão do gênero Klossinemella Costa, 1961 (Nematoda, Cobboldinidae), com descrição de uma nova subfamília e de uma nova espécie. Memórias do Instituto Oswaldo Cruz, 66, 169–179. https://doi.org/10.1590/S0074-02761968000200004
Gibbons, L. M., Khalil, L. F., & Marinkelle, C. J. (1995). New nematode genus, Podocnematractis, for Atractis ortleppi Thapar, 1925 (Cosmocercoidea: Atractidae) and the description of another new species, P. colombiaensis from turtles Podocnemis spp. in Colombia. Systematic Parasitology, 30, 47–56. https://doi.org/10.1007/BF00009244
Gibbons, L. M., Khalil, L. F., & Marinkelle, C. J. (1997). Paraorientatractis semiannulata n. g., n. sp. (Cosmocercoidea: Atractidae) from the Large Intestine of the Side-Necked Turtle, Podocnemis unifilis Troschel, 1848 (Testudines: Pelomedusidae) in Brazil. Memórias do Instituto Oswaldo Cruz, 92, 359–364. https://doi.org/10.1590/S0074-02761997000300011
Gibbons, M. L., & Platt, T. R. (2006). Rhinoclemmysnema n. g. and three new species of nematodes of the family Atractidae (Cosmocercoidea), with notes on the helminth fauna of Rhinoclemmys pulcherrrima (Testudines: Bataguridae) in Costa Rica. Journal of Helminthology, 80, 333–340. https://doi.org/10.1017/JOH2006360
González-Solís, D., & Mariaux, J. (2017). Orientatractis brycini sp. nov. (Nematoda: Atractidae) from characiform freshwater fishes in Gabon, Africa. Revue Suisse de Zoologie, 124, 1–8. https://doi.org/10.5281/zenodo.322659
González-Solís, D., & Moravec, F. (2004). Two new nematode species, Orientatractis campechensis n. sp. and Orientatractis chiapasensis n. sp. (Nematoda: Atractidae) from cichlid fishes in southern Mexico and Nicaragua. Journal of Parasitology, 90, 1443–1449. https://doi.org/10.1645/GE-3366
Khalil, L. F., & Gibbons, L. M. (1988). Two nematodes, Paratractis hystrix (Diesing, 1851) and Buckleyatractis marinkelli n. g., n. sp. (Atractidae: Cosmocercoidea) from Podocnemis spp. in Colombia. Systematic Parasitology, 12, 187–198. https://doi.org/10.1007/BF00007767
Leão, S. P. D., Famelli, S., & Vogt, R. C. (2019). Home range of Yellow-Spotted Amazon River Turtles (Podocnemis unifilis) (Testudines: Podocnemididae) in the Trombetas River Biological Reserve, Pará, Brazil. Chelonian Conservation and Biology, 18, 10–18. https://doi.org/10.2744/CCB-1273.1
Liu, B., Li, Z., & Zhang, L. (2018). A new species of Orientatractis (Nematoda: Atractidae) from the tortoise Indotestudo elongate (Blyth) in China. Acta Parasitologica, 63, 814–818. http://doi.org/10.1515/ap-2018-0099
Moravec, F., Kamchoo, K., & Pachanawan, A. (2015). New nematode species, Orientatractis mekongensis n. sp. (Atractidae) and Neosynodontisia suratthaniensis n. g., n. sp. (Pharyngodonidae) from freshwater fishes in Thailand. Systematic Parasitology, 92, 197–209. https://doi.org/10.1007/s11230-015-9598-4
Petter, A. J. (1966). Equilibre des espèces dans les populations de Nématodes parasites du colon des Tortues terrestres. Mémoires du Muséum National d’Histoire Naturelle, Series A., Zoologie, 39, 1–245.
Sarmiento, L. (1959). Description of Paratractis hystrix (Diesing, 1851) gen. nov. (Nematoda: Atractidae) from Podocnemis dumeriliana. The Journal of Parasitology, 45, 65–68. http://doi.org/10.2307/3274788