Paleodistribution modelling for planning the growth of natural history collections
Lázaro Guevara
Departamento de Zoología, Instituto de Biología, Universidad Nacional Autónoma de México, Apartado postal 70-153, 04510 Ciudad de México, Mexico
*Corresponding author: llg@ib.unam.mx (L. Guevara)
Abstract
This note aims to highlight the use of ecological niche models and paleodistributions (or past distributions) as a framework for planning surveys and natural history collection growth based on spatio-temporal hypotheses. I discuss why the usefulness of considering paleodistributions goes beyond suggesting areas that could harbor unknown records, by helping to identify biodiversity data gaps that can be crucial to studying evolutionary and ecological processes. This framework would allow for well-planned growth of collections and stimulate future long-term and multidisciplinary evolutionary research.
Keywords: Fieldwork; Niche modeling; Scientific collections; Quaternary
© 2019 Universidad Nacional Autónoma de México, Instituto de Biología. Este es un artículo Open Access bajo la licencia CC BY-NC-ND
Modelado de paleodistribuciones para planificar el crecimiento de colecciones de historia natural
Resumen
Esta nota tiene como objetivo resaltar el uso de modelos de nichos ecológicos y paleodistribuciones (o distribuciones pasadas) como un marco para la planificación del trabajo de campo y del crecimiento de museos de historia natural con base en hipótesis espacio-temporales. Discuto por qué la utilidad de considerar paleodistribuciones va más allá de sugerir áreas que podrían albergar registros desconocidos, ayudando a identificar brechas en los datos sobre la biodiversidad que pueden ser cruciales para estudiar procesos evolutivos y ecológicos. Este marco permitiría un crecimiento bien planificado de las colecciones y estimularía futuras investigaciones evolutivas multidisciplinarias a largo plazo.
Palabras clave: Trabajo de campo; Modelado de nichos; Colecciones científicas; Cuaternario
© 2019 Universidad Nacional Autónoma de México, Instituto de Biología. This is an open access article under the CC BY-NC-ND license
Introduction
Collecting and preserving specimens has been a crucial cornerstone in furthering our understanding of the natural world (Bradley et al., 2014; Holmes et al., 2016; Rowe et al., 2011; Winker, 2004). The benefits that scientific collecting and natural history collections provide to science and society are beyond debate (Graham et al., 2004; Suárez & Tsutsui, 2004). Even so, cutting the budgets for field-based research, scientific collecting, and maintaining and growing collections is becoming more frequent around the world (Kemp, 2015). Increasing the scientific collections is therefore a challenging issue, whose resolution will require new creativity for justifying and planning scientific collecting (Cook & Light, 2019). This is especially crucial in biodiversity-rich regions of the planet where knowledge gaps, threats to biodiversity, and funding limitations predominate (Dunnum et al., 2018).
Here, I show the use of ecological niche models and paleodistributions (or past distributions) as a framework for planning fieldwork and natural history collection growth based on spatio-temporal hypotheses. To do so, I emphasize why I consider that the usefulness of considering paleodistributions goes beyond suggesting areas that could harbor new records to increase scientific collections, by helping to identify biodiversity data gaps that can be crucial to studying evolutionary and ecological processes. This strategy would allow for well-planned growth of collections and stimulate future long-term evolutionary research.
Importantly, this contribution makes no attempt to debate the ethical aspects of scientific collecting, which have been dealt with extensively in the literature (Patterson, 2002; Sikes et al., 2011). Rather, I consider that acquiring necessary scientific knowledge about the biodiversity of our planet requires that natural history collections continue to grow through scientific collecting, and that this venture must continue to be supported by the scientific community and funding agencies (Cook & Light, 2019; Gippoliti, 2018).
Identifying data gaps and research needs
One priority of natural history museums is to capture the widest genetic and phenotypic variability in their collections as possible since an objective study of variability (e.g., genetic adaptations and phenotypic plasticity) is only possible through adequate geographic sampling (Gippoliti, 2018). An adequate geographical coverage within a scientific collection allows for a better understanding of the response of the species to past pressures such as climate change and the identification of populations that could assume more distinct ecological roles in the face of current global change (Ceballos & Ehrlich, 2009 .).
However, given that 1) genetic and phenotypic variability is not distributed homogeneously throughout the distribution area of a species, and 2) biological sampling is focused toward more easily accessible areas, it is inevitable that the current collections represent only a biased sample of the variability existing in nature (Illoldi-Rangel et al., 2004; Jarnevich et al., 2015; Meineke et al., 2018; Winker, 2004; Zamudio et al., 2016). This may be the case even for species that are well-represented in terms of the number of specimens in collections. Predicting where we might find natural variability that is not yet preserved in collections would be very helpful for making decisions about what and where to collect, allowing better use of shrinking resources.
Niche modelling and paleodistributions
Ecological -correlative- Niche Modeling (ENM) estimates the current potential species distribution by identifying geographical regions with environmental conditions similar to those where the presence of the species has been confirmed (Soberón & Peterson, 2005). Among its numerous applications (for an example, see Araújo et al., 2019), ENM can be used to identify regions on the planet, mainly those that are remote and poorly sampled, that could harbor yet unknown components of biodiversity (e.g., new populations of known species, or even species that are not yet known; Raxworthy et al., 2003).
Niche models can also be projected onto Quaternary climates simulated with Global Circulation Models to estimate the potential distribution of species through time (i.e., paleodistributions; Peterson & Lieberman, 2012). For example, ENM have been used to estimate geographic paleodistributions through different epochs from the Last Glacial Maximum (ca. 21,000 years) to the present, in order to track historical species distributions under different climates (Nogués-Bravo, 2009; Peterson & Lieberman, 2012). Specifically, paleodistributions through the last glacial-interglacial cycle are relevant in evolutionary biology, since this time period has been a causal factor in current geographical distribution patterns. The abrupt alternation between hot (even warmer than the present) and cold periods is thought to have caused expansion/contraction and fragmentation/historical connectivity events that should leave signals on the distributions of current lineages; it is therefore theoretically possible to predict the expected genetic and phenotypic variability of known or still unknown lineages (Blois et al., 2010; Chan et al., 2011; Hewitt, 2000; Zamudio et al., 2016).
Beyond new records
To illustrate the utility of paleodistribution modelling for planning the growth of natural history collections, I used the small-eared shrews of the Cryptotis mexicanus group (Mammalia, Eulipotyphla) as an example. This group is mainly distributed in the tropical montane cloud forest of Mexico (Cryptotis magnus, C. mexicanus, C. nelsoni, and C. phillipsii). It is a relatively well-known clade, with a large number of specimens in collections and with georeferenced localities (Guevara et al., 2015; Guevara & Sánchez-Cordero, 2018). Using maxent, an open source software for modeling species distributions (Phillips et al., 2017), the ecological niche of this clade was estimated to predict its potential distribution at present and at the following times of the last glacial-interglacial cycle under the CCSM3 climate scenario (Community Climate System Model; Collins et al., 2006): Late-Holocene (4.2 – 0.3 ka), Mid-Holocene (8.326 – 4.2 ka), Early-Holocene (11.7 – 8.326 ka), Younger Dryas Stadial (12.9-11.7 ka), Bølling-Allerød Interstadial (14.7-12.9 ka), Heinrich Stadial 1 (17.0-14.7 ka), and Last Glacial Maximum (ca. 21 ka) (Fig. 1).
By superimposing the records of the Cryptotis mexicanus group that are currently available in natural history collections, it is possible not only to detect regions that could harbor new records, but also to identify if they come from areas of postglacial colonization, areas of predicted stability (refugial zones), and if they have maintained contact that could allow gene flow within this group of species (Chan et al., 2011).
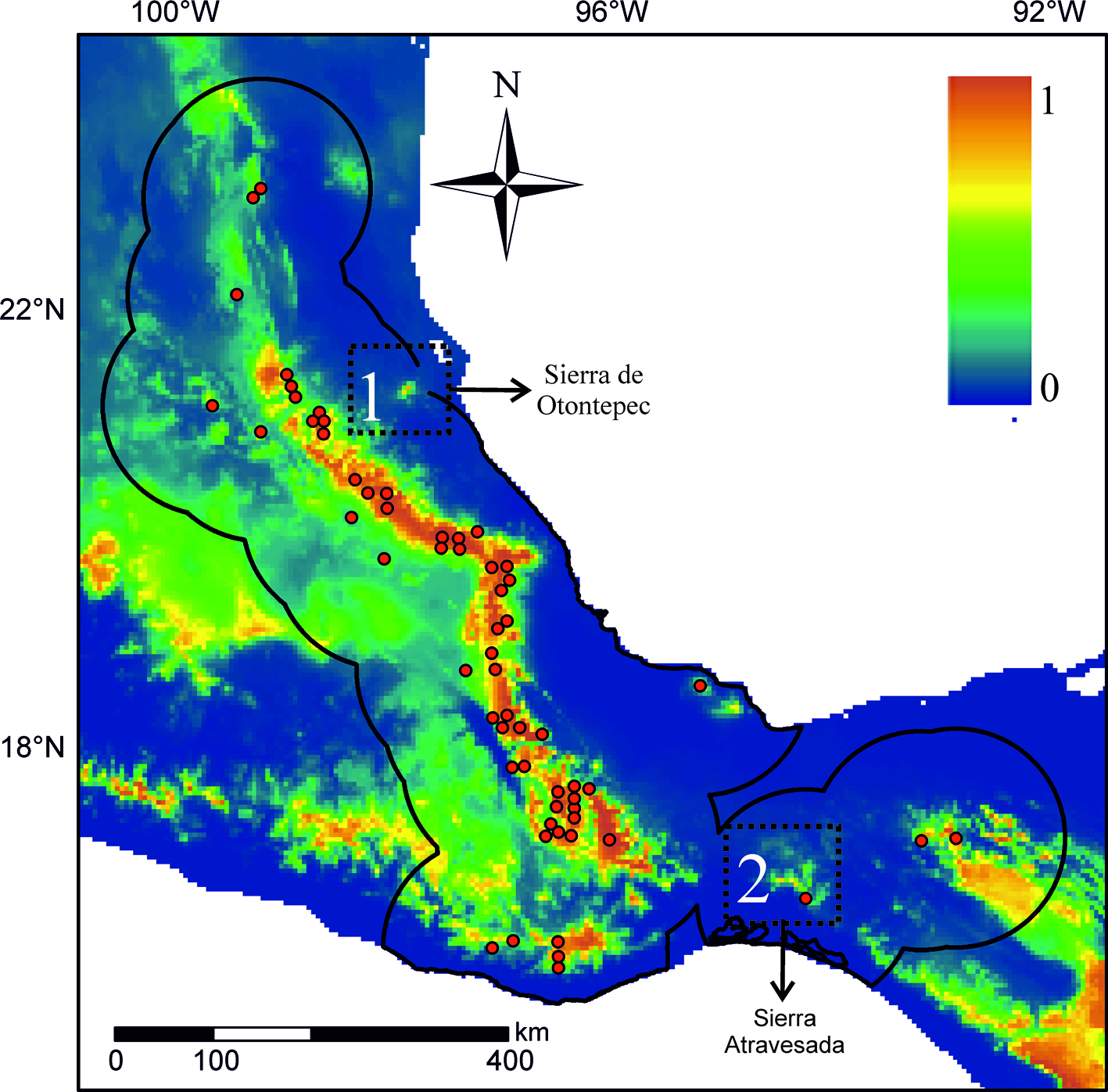
As an example, I highlight 2 relatively poorly sampled regions for small mammals in Mexico (Briones-Salas & Sánchez-Cordero, 2004; Guevara & Sánchez-Cordero, 2018; Fig. 2). According to the paleodistribution hypothesis, region 1 or Sierra de Otontepec has been climatically suitable for the C. mexicanus group through the last glacial-interglacial cycle, so it is reasonable to predict the existence of new populations. Notably, a recent expedition found 6 individuals of C. mexicanus in this region (Mayen-Zaragoza et al., 2019). Going one step beyond new records, we could also predict this population to be the result of postglacial colonization from the mountain range to the west, specifically during the Heinrich Stadial 1.
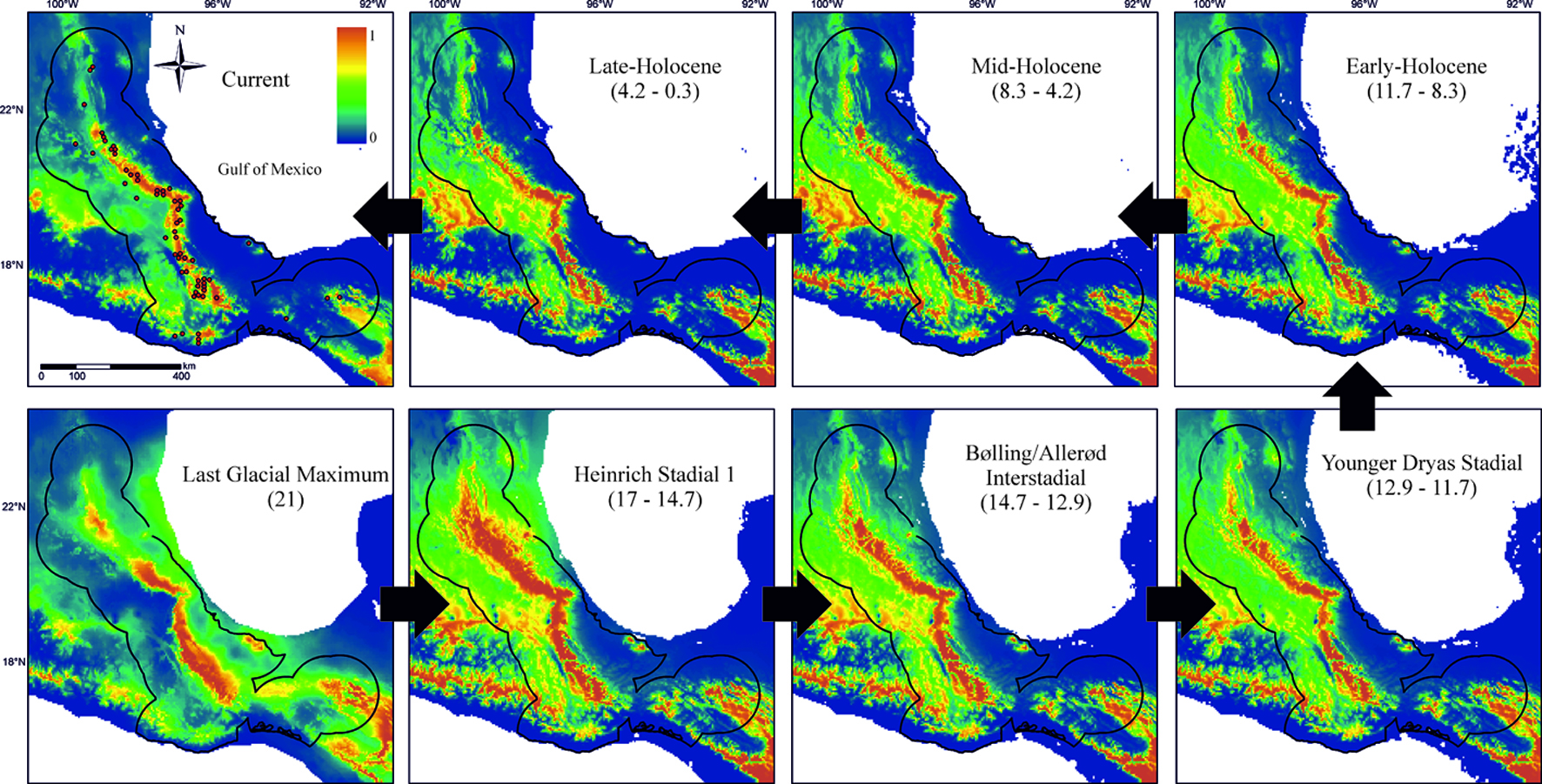
Region 2 borders the “Sierra Atravesada” that lies in the middle of the Isthmus of Tehuantepec, which represents a long-standing biogeographical dilemma about its role as barrier for montane species (Peterson et al., 1999). The “Sierra Atravesada” has a record supported by a single individual collected more than 50 years ago and for which no tissue samples are available (California Academy of Science, CAS 14636). Based on the paleodistribution hypothesis, this region did not have adequate climatic conditions for the group C. mexicanus during the Last Glacial Maximum, but the populations had to move north and formed a bridge along the Isthmus, promoting gene flow between populations on either side of the Isthmus. We could then expect that this population is not highly differentiated from the populations on either side of the Isthmus. The 2 examples described here can be later tested through obtaining more specimens or samples and using genetic and morphological analyses.
I aim to illustrate using this simple example that paleodistributions have the potential to provide information about the causes or events that could explain current distributional patterns, as well as to predict patterns of genetic and phenotypic variability of still-unknown biodiversity. The framework outlined here can certainly be applied to questions on broader taxonomic and geographic scales. For example, including more species with similar ecological characteristics could provide information about the dynamics of communities over time. This example also shows that even for relatively well-known areas and taxa, there are both gaps in knowledge and opportunities for field-based research that if addressed could greatly enrich our integrated understanding of the evolution, ecology, and conservation of biodiversity.
Perspective
The challenges that humanity faces require that collections continue to grow. However, they do not just need the preservation of more specimens per se, but the preservation of more historical events that allow us to better understand complex biological processes at the species and community levels (Schindel & Cook, 2018). The framework presented in this opinion is a proposal, based on spatio-temporal hypotheses, for the kind of planning that should go into collections. As a parallel step, these specimens or samples collected following this framework will serve to continue the validation of ecological niche models and paleodistributions, a task that urgently needs to be expanded and improved (Araújo et al., 2019). I hope this opinion also serves to stimulate more integrated and cooperative long-term research among niche modelers, taxonomists, ecologists, and biogeographers, or, as Futuyma (1998) said, the creation of future scientific naturalists.
Acknowledgments
To C. Granados, A. Moreno, and P. Vélez for their invitation to teach a seminar at the Institute of Biology of the UNAM, which is part of this opinion note. I would like to thank the two anonymous reviewers for their valuable comments to improve the quality of this opinion.
References
Araújo, M. B., Anderson, R. P., Barbosa, A. M., Beale, C. M., Dormann, C. F., Early, R. et al. (2019). Standards for distribution models in biodiversity assessments. Science Advances, 5, eaat4858. https://doi.org/10.1126/sciadv.aat4858
Barve, N., Barve, V., Jiménez-Valverde, A., Lira-Noriega, A., Maher, S. P., Peterson, A. T. et al. (2011). The crucial role of the accessible area in ecological niche modeling and species distribution modeling. Ecological Modelling, 222, 1810–1819. https://doi.org/10.1016/j.ecolmodel.2011.02.011
Blois, J. L., McGuire, J. L., & Hadly, E. A. (2010). Small mammal diversity loss in response to late-Pleistocene climatic change. Nature, 465, 771–774. https://doi.org/10.1038/nature09077
Bradley, R. D., Bradley, L. C., Garner, H. J., & Baker, R. J. (2014). Assessing the value of natural history collections and addressing issues regarding long-term growth and care. BioScience, 64, 1150–1158. https://doi.org/10.1093/biosci/biu166
Briones-Salas, M., & Sánchez-Cordero, V. (2004). Mamíferos. Biodiversidad de Oaxaca. México D.F.: Instituto de Biología, Universidad Nacional Autónoma de México/ Fondo Oaxaqueño para la Conservación de la Naturaleza/ World Wildlife Fundation.
Ceballos, G., & Ehrlich, P. R. (2009). Discoveries of new mammal species and their implications for conservation and ecosystem services. Proceedings of the National Academy of Sciences, 106, 3841–3846. https://doi.org/10.1073/pnas.0812419106
Chan, L. M., Brown, J. L., & Yoder, A. D. (2011). Integrating statistical genetic and geospatial methods brings new power to phylogeography. Molecular Phylogenetics and Evolution, 59, 523–537. https://doi.org/10.1016/j.ympev.2011.01.020
Collins, W. D., Bitz, C. M., Blackmon, M. L., Bonan, G. B., Bretherton, C. S., Carton, C. et al. (2006). The community climate system model version 3 (CCSM3). Journal of Climate, 19, 2122–2143. https://doi.org/10.1175/JCLI3761.1
Cook, J. A., & Light, J. E. (2019). The emerging role of mammal collections in 21st century mammalogy. Journal of Mammalogy, 100, 733–750. https://doi.org/10.1093/jmammal/gyy148
Dunnum, J. L., McLean, B. S., & Dowler, R. C. (2018). Mammal collections of the Western Hemisphere: a survey and directory of collections. Journal of Mammalogy, 99, 1307–1322. https://doi.org/10.1093/jmammal/gyy151
Futuyma, D. J. (1998). Wherefore and whither the naturalist? The American Naturalist, 151, 1–6. https://doi.org/10.1086/286097
Gippoliti, S. (2018). Natural history collecting and the arrogance of the modern Ark researcher. Bionomina, 13, 69–73. http://dx.doi.org/10.11646/bionomina.13.1.6
Graham, C. H., Ferrier, S., Huettman, F., Moritz, C., & Peterson, A. T. (2004). New developments in museum-based informatics and applications in biodiversity analysis. Trends in Ecology & Evolution, 19, 497–503. https://doi.org/10.1016/j.tree.2004.07.006
Guevara, L., Cervantes, F. A., & Sánchez-Cordero, V. (2015). Riqueza, distribución y conservación de los topos y las musarañas (Mammalia, Eulipotyphla) de México. Therya, 6, 43–68. http://dx.doi.org/10.12933/therya-15-211
Guevara, L., & Sánchez-Cordero, V. (2018). Patterns of morphological and ecological similarities of small-eared shrews (Soricidae, Cryptotis) in tropical montane cloud forests from Mesoamerica. Systematics and Biodiversity, 16, 551–564. https://doi.org/10.1080/14772000.2018.1470582
Hewitt, G. (2000). The genetic legacy of the Quaternary ice ages. Nature, 405, 907–913. https://doi.org/10.1038/35016000
Holmes, M. W., Hammond, T. T., Wogan, G. O., Walsh, R. E., LaBarbera, K., Wommack, E. A. et al. (2016). Natural history collections as windows on evolutionary processes. Molecular Ecology, 25, 864–881. https://doi.org/10.1111/mec.13529
Illoldi-Rangel, P., Sánchez-Cordero, V., & Peterson, A. T. (2004). Predicting distributions of Mexican mammals using ecological niche modeling. Journal of Mammalogy, 85, 658–662. https://doi.org/10.1644/BER-024
Jarnevich, C. S., Stohlgren, T. J., Kumar, S., Morisette, J. T., & Holcombe, T. R. (2015). Caveats for correlative species distribution modeling. Ecological Informatics, 29, 6–15. https://doi.org/10.1016/j.ecoinf.2015.06.007
Kemp, C. (2015). Museums: the endangered dead. Nature News, 518, 292–294. https://doi.org/10.1038/518292a
Mayén-Zaragoza, M., Guevara, L., Hernández-Canchola, G., & León-Paniagua, L. (2019). First record of shrews (Eulipotyphla, Soricidae) in the Sierra de Otontepec, an isolated mountain in Veracruz, Mexico. Therya, 10, 59–63. https://doi.org/10.12933/therya-19-690 ISSN 2007-3364
Meineke, E. K., Davies, T. J., Daru, B. H., & Davis, C. C. (2018). Biological collections for understanding biodiversity in the Anthropocene. Philosophical Transactions of the Royal Society B, 374, 20170386. https://doi.org/10.1098/rstb.2017.0386
Nogués-Bravo, D. (2009). Predicting the past distribution of species climatic niches. Global Ecology and Biogeography, 18, 521–531. https://doi.org/10.1111/j.1466-8238.2009.00476.x
Patterson, B. D. (2002). On the continuing need for scientific collecting of mammals. Mastozoología Neotropical, 9, 253–262.
Peterson, A. T., & Lieberman, B. S. (2012). Species’ geographic distributions through time: playing catch-up with changing climates. Evolution: Education and Outreach, 5, 569–581. https://doi.org/10.1007/s12052-012-0385-2
Peterson, A. T., Soberón, J., & Sánchez-Cordero, V. (1999). Conservatism of ecological niches in evolutionary time. Science, 285, 1265–1267. https://doi.org/10.1126/science.285.5431.1265
Phillips, S. J., Anderson, R. P., Dudík, M., Schapire, R. E, & Blair, M. E. (2017). Opening the black box: an open-source release of Maxent. Ecography, 40, 887–893. https://doi.org/10.1111/ecog.03049
Raxworthy, C. J., Martínez-Meyer, E., Horning, N., Nussbaum, R. A., Schneider, G. E., Ortega-Huerta, M. A. et al. (2003). Predicting distributions of known and unknown reptile species in Madagascar. Nature, 426, 837–841. https://doi.org/10.1038/nature02205
Rowe, K. C., Singhal, S., Macmanes, M. D., Ayroles, J. F., Morelli, T. L., Rubidge, E. M. et al. (2011). Museum genomics: low-cost and high-accuracy genetic data from historical specimens. Molecular Ecology Resources, 11, 1082–1092. https://doi.org/10.1111/j.1755-0998.2011.03052.x
Schindel, D. E., & Cook, J. A. (2018). The next generation of natural history collections. Plos Biology, 16, e2006125. https://doi.org/10.1371/journal.pbio.2006125
Sikes, R. S., Gannon, W. L., & The Animal Care and Use Committee of the American Society of Mammalogists. (2011). Guidelines of the American Society of Mammalogists for the use of wild mammals in research. Journal of Mammalogy, 92, 235–253. https://doi.org/10.1644/10-MAMM-F-355.1
Soberón, J., & Peterson, A. T. (2005). Interpretation of models of fundamental ecological niches and species’ distributional areas. Biodiversity Informatics, 2, 1–10. https://doi.org/10.17161/bi.v2i0.4
Suárez, A. V., & Tsutsui, N. D. (2004). The value of museum collections for research and society. AIBS Bulletin, 54, 66–74. https://doi.org/10.1641/0006-3568(2004)054[0066:TVOMCF]2.0.CO;2
Winker, K. (2004). Natural history museums in a postbiodiversity era. AIBS Bulletin, 54, 455–459. https://doi.org/10.1641/0006-3568(2004)054[0455:NHMIAP]2.0.CO;2
Zamudio, K. R., Bell, R. C., & Mason, N. A. (2016). Phenotypes in phylogeography: Species’ traits, environmental variation, and vertebrate diversification. Proceedings of the National Academy of Sciences, 113, 8041–8048. https://doi.org/10.1073/pnas.1602237113
